

| Online: | |
| Visits: | |
| Stories: |

| Story Views | |
| Now: | |
| Last Hour: | |
| Last 24 Hours: | |
| Total: | |
Cheap Efficient Catalyst Developed to Replace Platinum for Hydrogen Production
Platinum is currently the most efficient catalytic electrode material for generating hydrogen in this way, but the precious metal is both scarce and expensive. Yee-Fun Lim and colleagues at the A*STAR Institute of Materials Research and Engineering have now developed electrocatalyst nanoparticles that are highly active, cheap and stable, and which perform the hydrogen evolution reaction as well as any alternatives to platinum yet discovered.
© 2017 A*STAR Institute of Materials Research and Engineering
During the reaction, electricity helps metal atoms on the surface of these nanoparticles to pluck a hydrogen atom from a water molecule. The hydrogen atom then combines with another hydrogen atom — either on the nanoparticle’s surface, or from another water molecule — to make hydrogen gas (H2). Crucially, the metal sulfide nanoparticles operate well under the alkalineconditions usually required for the parallel reaction that generates oxygen during water splitting.
Lim’s team showed that varying the temperature and duration of the heating step used to prepare the nanoparticles had a dramatic effect on their composition and relative proportions, and tests showed that this determined their activity in the hydrogen evolution reaction. Prolonged heating caused some of the nanoparticles to clump together, for example, and also increased the proportion of cobalt sulfide, which significantly reduced the catalyst’s activity.
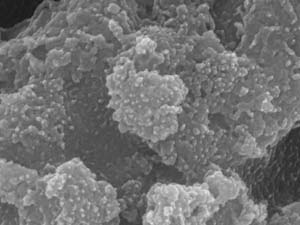
The best performance came from the mixed-metal sulfide that had been heated to 500 degrees Celsius for just 10 minutes (see image). It required a relatively low voltage of 163 millivolts to initiate the hydrogen evolution reaction, just 47 millivolts higher than a commercial platinum electrocatalyst, and comparable to the best alternatives. The catalyst showed no degradation over three days of continuous reactions.
“The mixed catalyst combines the good properties of both nickel and cobalt catalysts to achieve superior performance,” says Lim. His team plans to use a similar approach to tailor-make catalytic nanoparticles for a different reaction that turns carbon dioxide into fuels.
The A*STAR-affiliated researchers contributing to this research are from the Institute of Materials Research and Engineering. For more information about the team’s research, please visit the Ceramic Materials group webpage.
Contacts and sources:
The Agency for Science, Technology and Research (A*STAR)
Source: http://www.ineffableisland.com/2017/03/cheap-efficient-catalyst-developed-to.html


