| Online: | |
| Visits: | |
| Stories: |

| Story Views | |
| Now: | |
| Last Hour: | |
| Last 24 Hours: | |
| Total: | |
Intramolecular Hydrogen Bonds, We Hardly Knew Thee
Intramolecular hydrogen bonds (IHBs) are interesting beasts. They can be used to improve the potency of a drug by constraining it in a bioactive conformation, and they can be used to hide polarity and improve permeability; cyclosporin being the classic example of the latter.
But the exact amount of potency gain you get through formation of an intramolecular hydrogen bond is not clear. Conformationally you could constrain a molecule through an IHB, but you would still be introducing polarity and hydrogen bond donors and acceptors, and this will lead to some desolvation penalties that would have to be exactly compensated for by the IHB to lead to a net positive effect. There’s a group from D. E. Shaw and AstraZeneca who have now looked at about 1200 cases of matched molecular pairs and their biological activities to figure out the contribution of IHBs to potency. The average gain they see? Close to zero. This means that on average, an IHB is as likely to blunt your potency as it is to improve it.
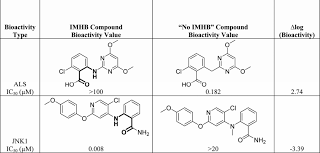
What’s more interesting are the outliers. A small but distinct fraction of results display matched molecular pairs in which there is a gain of at least 2 to 3 log units. Inspection of these results sheds light on why these cases really benefit from the formation of IHBs, but it’s also important to not overemphasize the positive role of the IHBs in every instance, especially in cases where the non-IHB matched pair displays particular severe repulsive interactions.
For instance there’s an N-Me vs N-H pair in which the latter can form an IHB while the former cannot. But not only can the former not form this bond, the N-Me probably causes a steric repulsion and causes a very different conformational profile, perhaps leading to a conformational penalty in binding to the target. Similarly in another case, the matched pair presents an N-H vs O difference. Here again, not only can the oxygen not form the bond, but it likely strongly repels the other oxygen participating in the IHB. Thus, these outliers are outliers not because the IHB is particularly stable, but because the complementary arrangement is particularly unstable.
Nonetheless, this is a nice study to keep in mind every time you want to use an IHB as a tactic for improving potency or permeability. It may well work, but then it may well not. As in most cases in drug discovery, the decision to incorporate an IHB-forming element will be dictated by many other factors including cost, resources and synthetic accessibility. As with many other tactics in the field, when it comes to IHBs, caveat emptor.
Source: http://wavefunction.fieldofscience.com/2017/04/intramolecular-hydrogen-bonds-we-hardly.html